Primary reverse osmosis device+EDIEquipment (pharmaceutical purified water equipment)
pharmaceuticalPurified WaterequipmentordinaryPharmaceutical purified water equipmentThe difference is that the entire system uses pasteurization to avoid any risk of chemical residues.
pharmaceuticalPurified WaterequipmentOptional disinfection methods: activated carbon pasteurizationCIP cleaning system, ozone sterilization for distribution system, pasteurization for distribution system, and pure steam sterilization for distribution system.
(1)pharmaceuticalPurified WaterequipmentDesign and manufacturing standards: The system is designed according to the requirements of the new Chinese, American, and European Pharmacopoeia standards, followingCGMP and GAMP standards comply with GMP and FDA certification requirements.
(2)Pharmaceutical purified water equipmentThe customer is fully involved, demonstrating the details and effects in advance to fully meet their needs.

(3)Pharmaceutical purified water equipmentAdopting modular design, it is divided into three modules: pre-processing module, host module, and distribution system. All three modules use pasteurization, pre-treatment, and the host system and distribution system use double tube plate and tube heat exchangers to disinfect activated carbon, reverse osmosis, and process pipelines. Do not use any chemical agents to avoid any chemical residues.
(4)Pharmaceutical purified water equipmentRealize full lifecycle data traceability, using world advanced paper-based or paperless recorders for data recording, usingUPS power supply can prevent system data loss caused by power outages.
(5)Pharmaceutical purified water equipment, usingThe CIP cleaning system and the pasteurization system share the same system to perform CIP cleaning and pasteurization on the system. Using high-temperature membrane components to achieve pasteurization function. Adopting a double tube plate tube heat exchanger that meets the standards for injection water and avoids the risk of water vapor cross contamination.
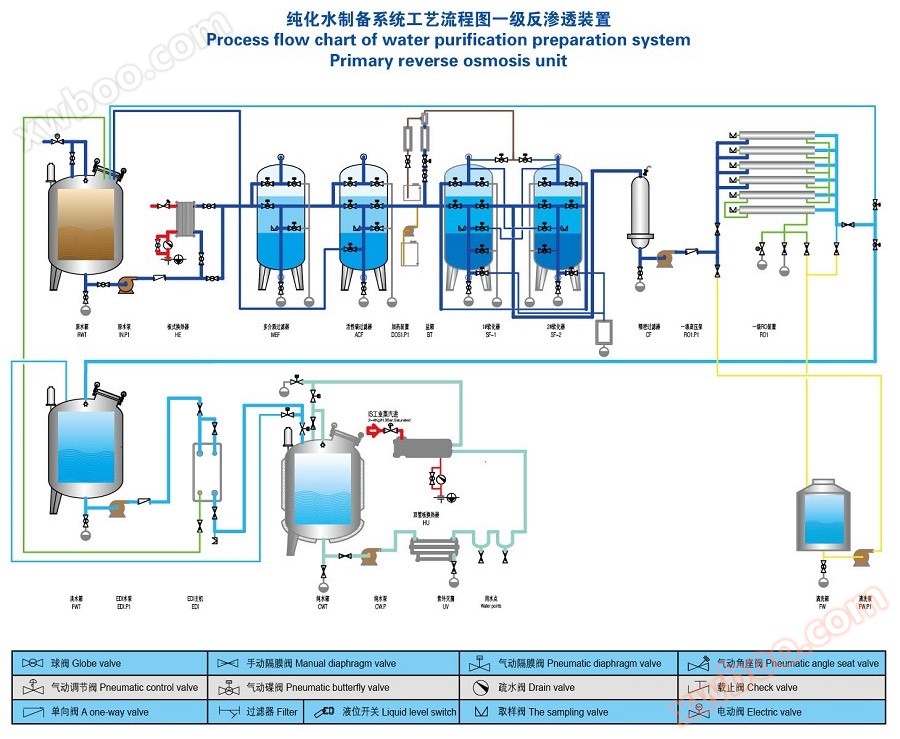
(6)Pharmaceutical purified water equipmentThe purified water distribution system adopts a modular design to achieve online monitoring and recording of key parameters such as flow rate, temperature, and conductivity. The system uses a double tube plate and tube heat exchanger for pasteurization. The purified water distribution system and the host system share a control system to avoid duplicate designs. The entire equipment follows a set of procedures to reduce operational risks for operators.

